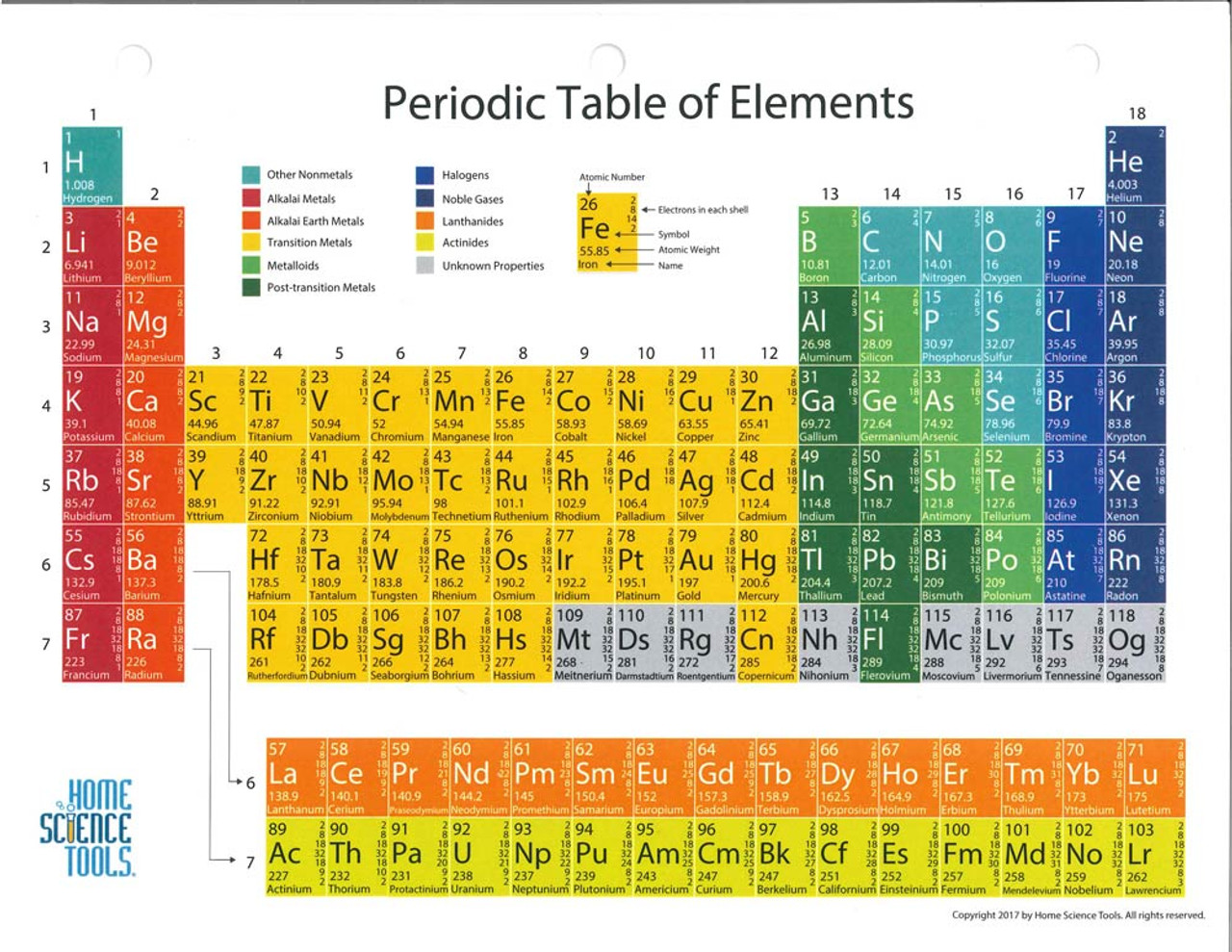
Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms. For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Staar chemistry reference sheet - chemistry reference sheet periodic table Chemistry reference sheet periodic table of the elements california standards test 1 1a 1 1 h hydrogen 1.01 3 2 li 18 8a 2 2 2a 4 9.01 11 12 na mg sodium magnesium 22.99 24.31 19 4 na beryllium 6.94 3 lithium k 20 ca sodium 22.99 3 3b 21 sc 4 4b. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. These periodic tables use accurate data for name, atomic number, element symbol, atomic weight, and electron configuration, obtained from the International Union of Pure and. These individual element summary pages contain a lot of additional. When exploring the table or list views on this page, please note the links to dedicated pages for each element. Some tables are available as slides in Google Apps. PubChem is providing this periodic table page in order to help navigate abundant chemical element data available in PubChem. Mendeleevs Periodic Table was published in the German Journal of chemistry. 
This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. 4 of 15 Periodic table of the elements 1 2 H 1.01 H 1.01 He 4.00 3 4 5 6 7 8 9 10 Li 6.94 Be 9.01 B 10.81 C 12.01 N 14.01 O 16.00 F 19.00 Ne 20.18 11 12 13 14 15 16. This is a collection of free printable periodic tables in PDF file or PNG image format to save, print, and use. Periodic Table of Elements (Interactive) PDF - This Interactive Periodic Table. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. Many high school students find that chemistry is a challenging subject for a variety of reasons. The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital. PDF Chemistry Reference Tables - North Carolina Public Schools.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
